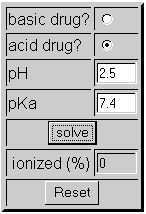
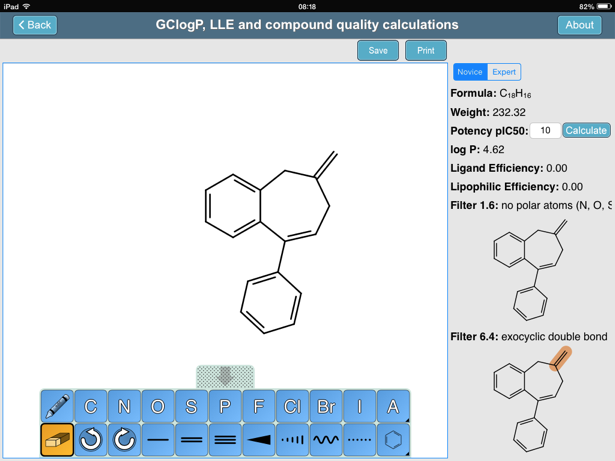
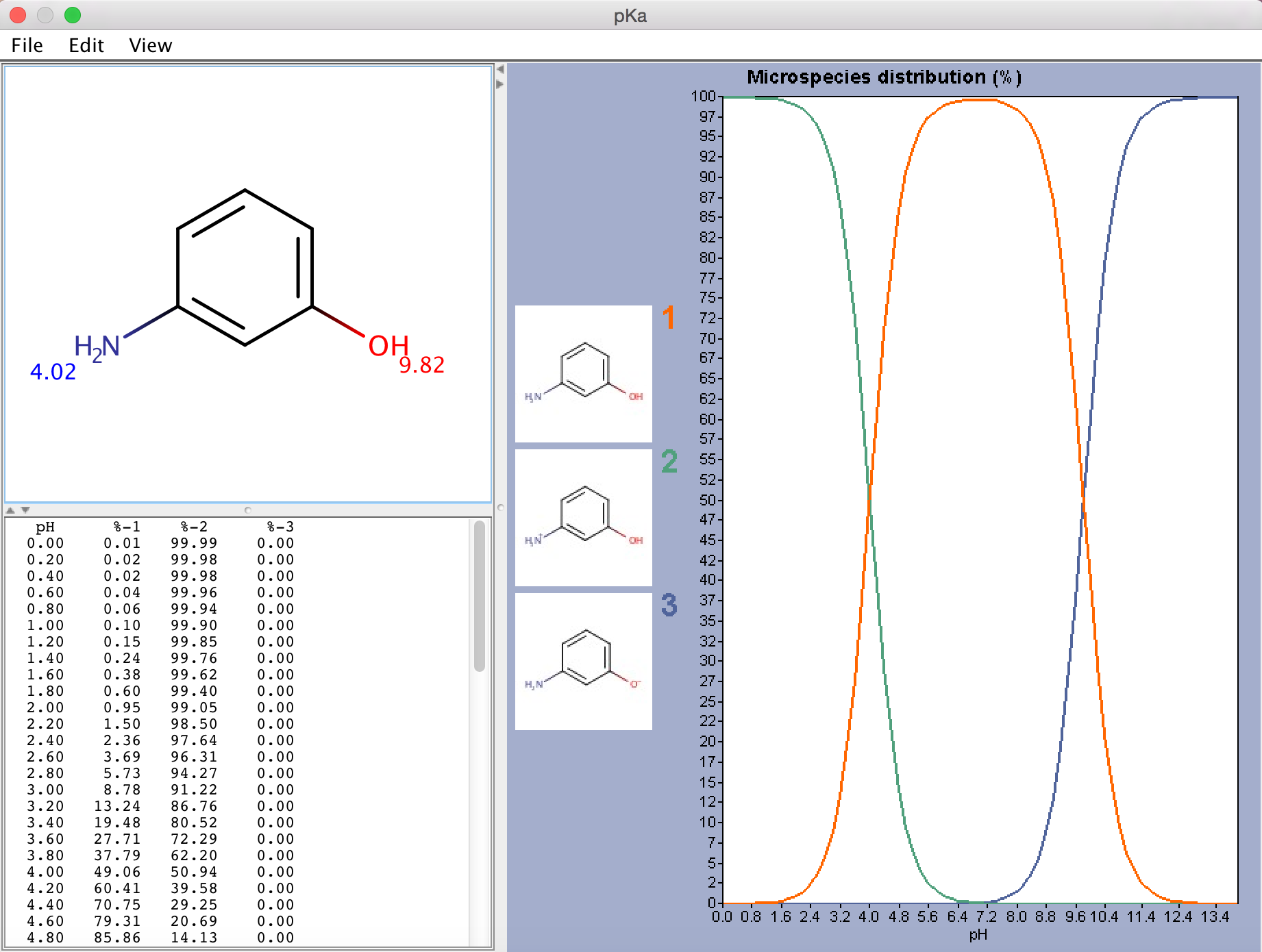
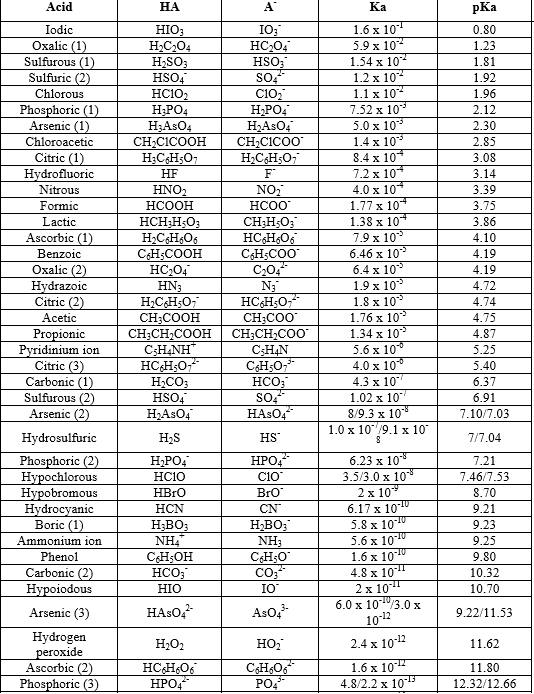
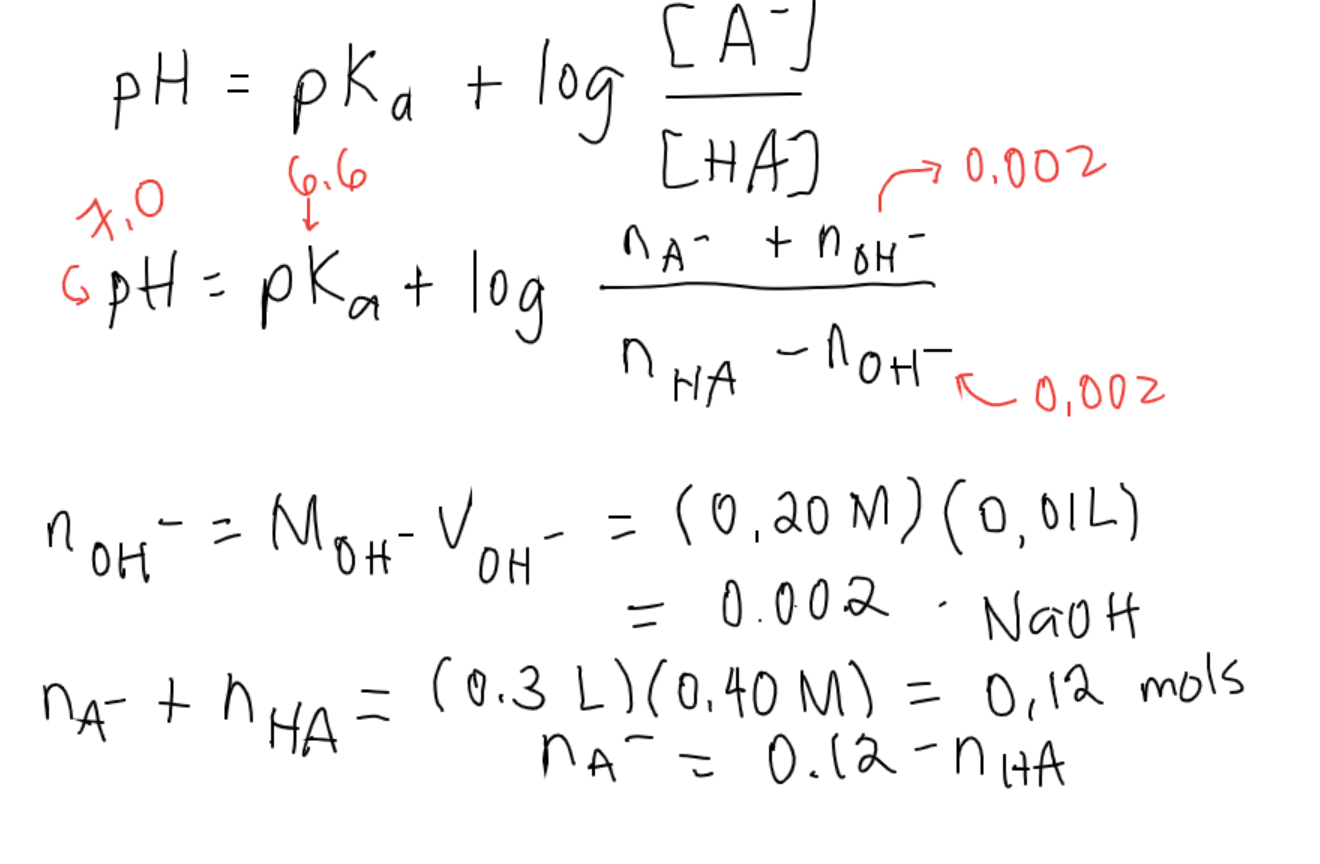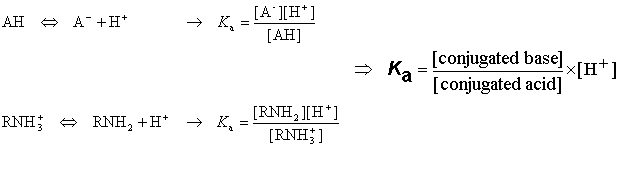How should the acid dissociation constant pKa be measured? | Automatic Potentiometric Titrators | Faq | Kyoto Electronics Manufacturing Co.,Ltd.("KEM")

SOLVED: pH = pKa + log10 Using the Henderson-Hasselbalch equation as seen above, solve for pKa. Given the mixture of 0.30 M sodium acetate and 0.20 M acetic acid, what is the

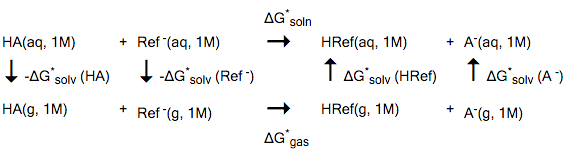
The thermodynamic cycles for calculation (from top to bottom): pKa, pKH... | Download Scientific Diagram

Density Functional Theory Calculation of pKa's of Thiols in Aqueous Solution Using Explicit Water Molecules and the Polarizable Continuum Model | The Journal of Physical Chemistry A