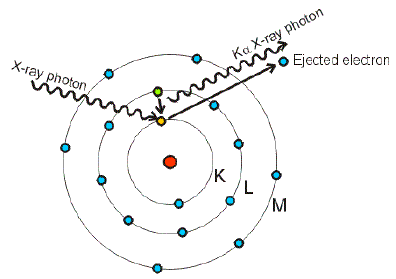
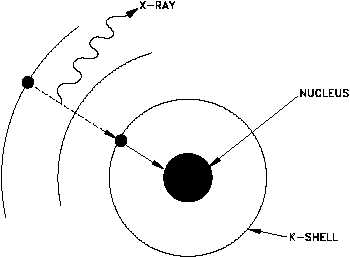
The energy of a K-electron in tungsten is -20 KeV and of an L-electron is -2 KeV. Find the wavelength of X- rays emitted when there is electron jump from L to
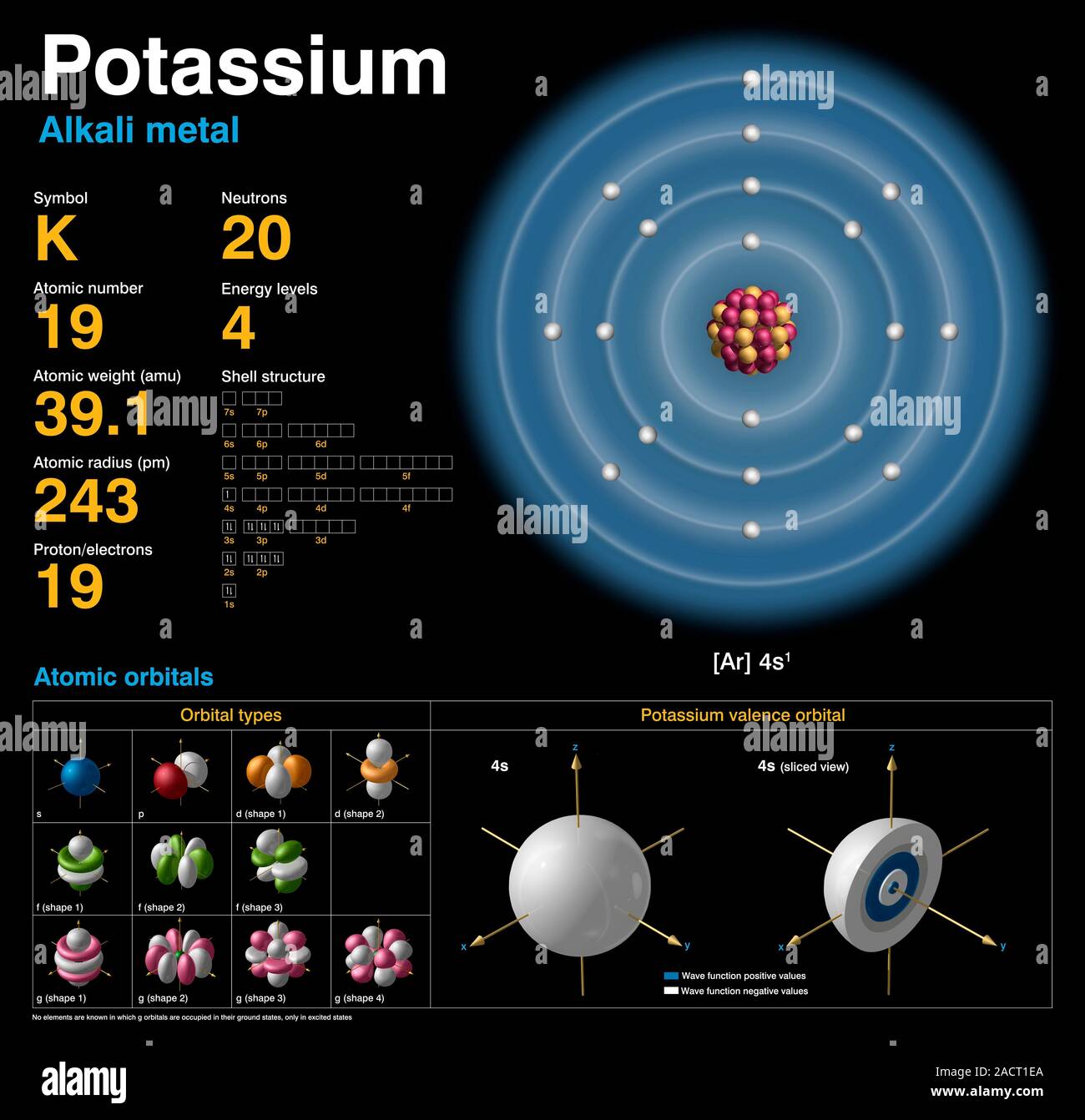
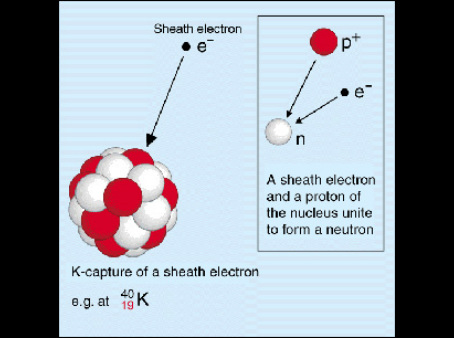
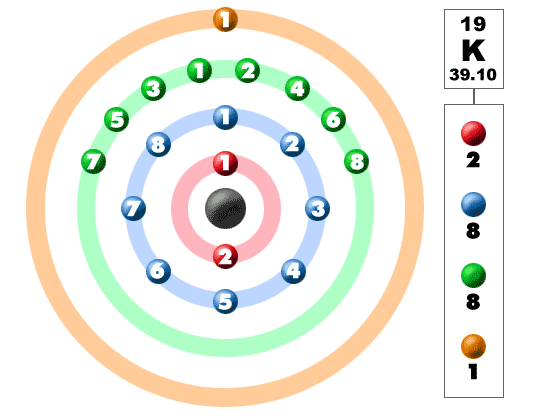
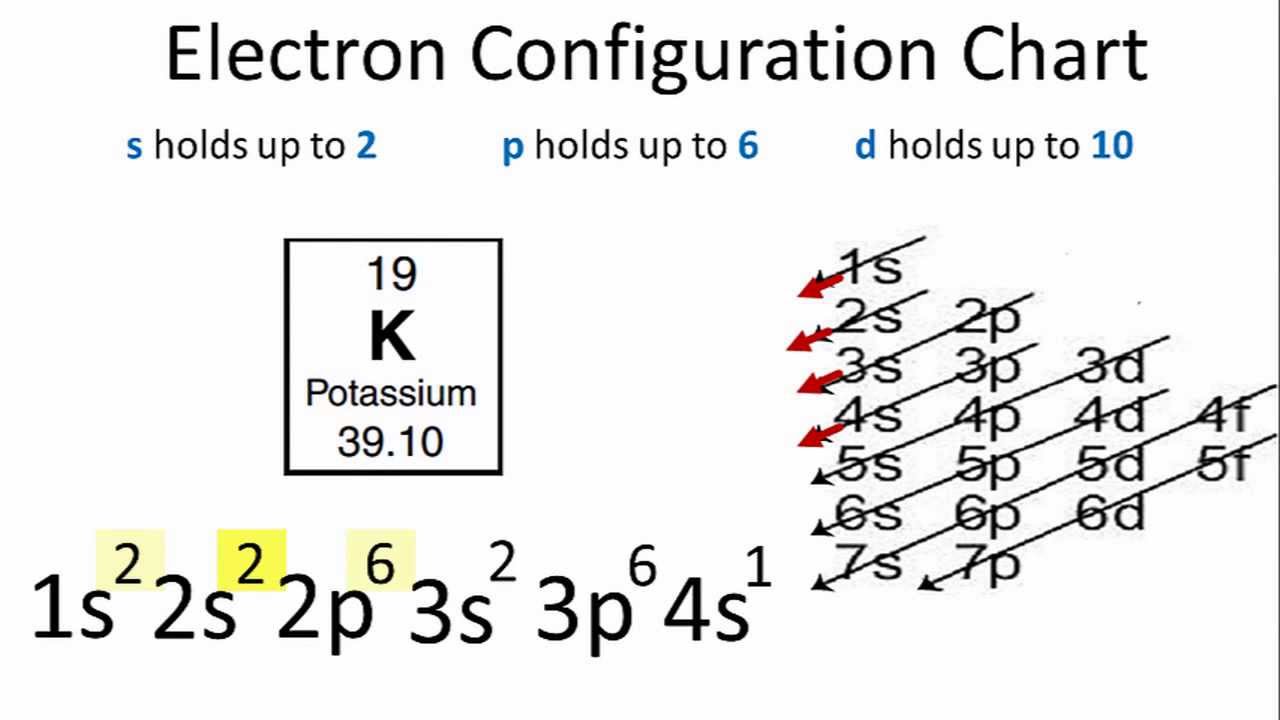
Potassium (K). Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of potassium-39 (atomic numb Stock Photo - Alamy