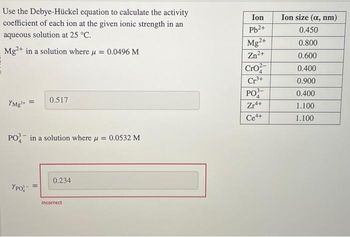
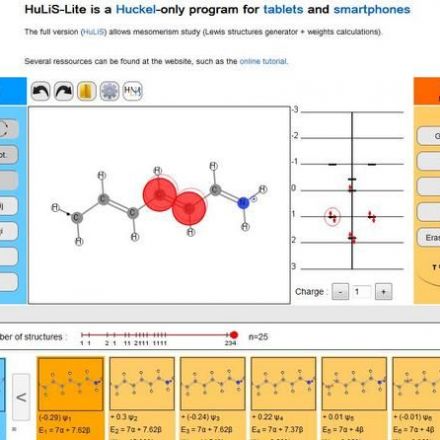
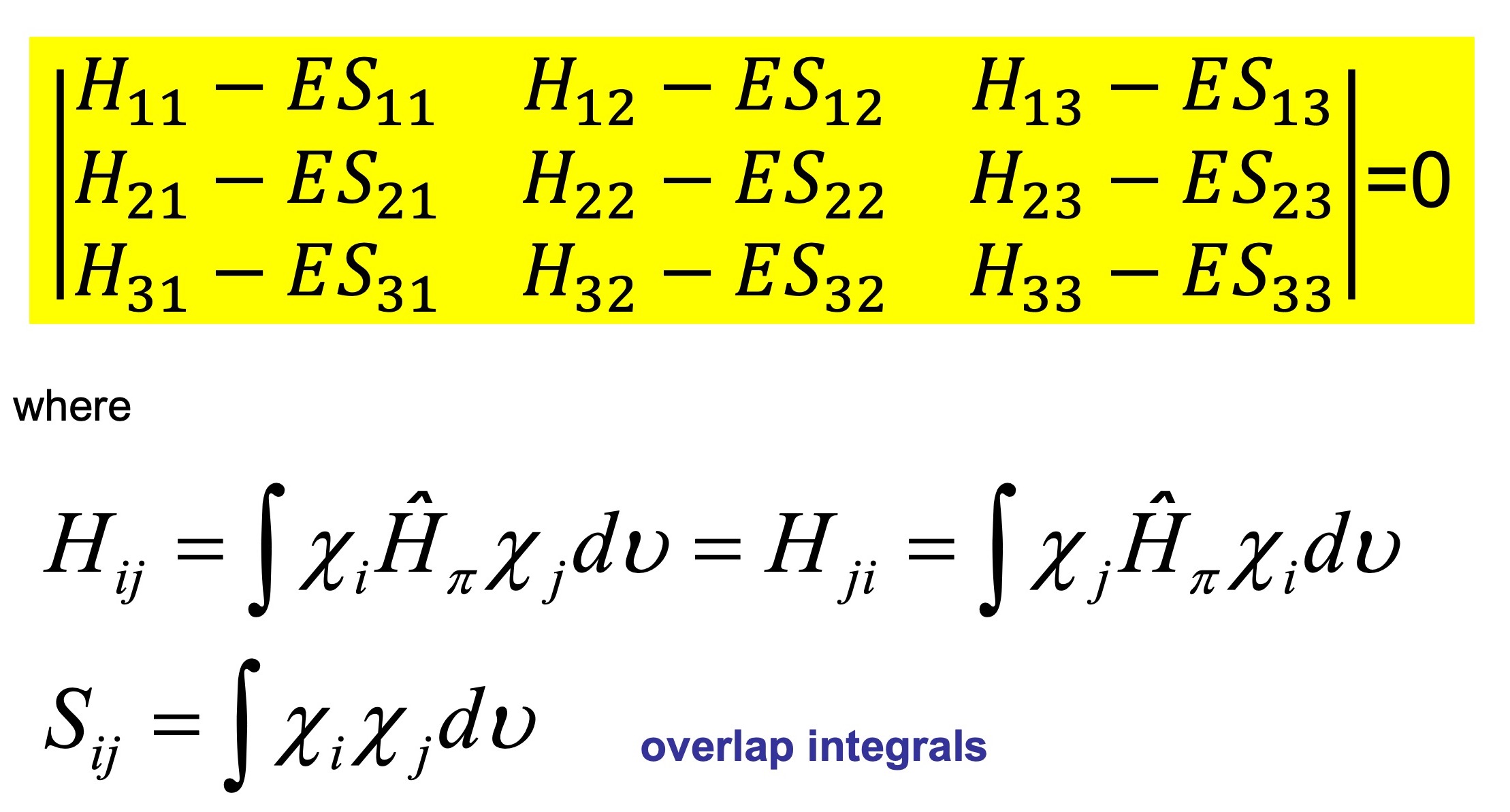SOLVED: Texts: Use the Debye-Huckel equation to calculate the activity coefficient of each ion at the given ionic strength in an aqueous solution at 25°C. Ion Ion size (nm) Pb2+ 0.450 Mg2+

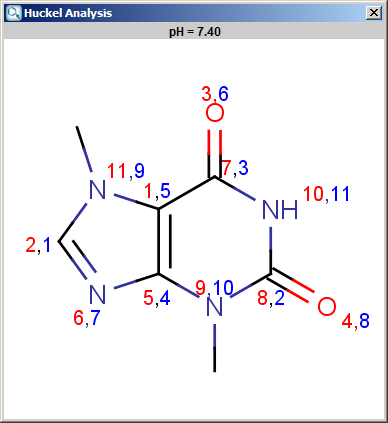
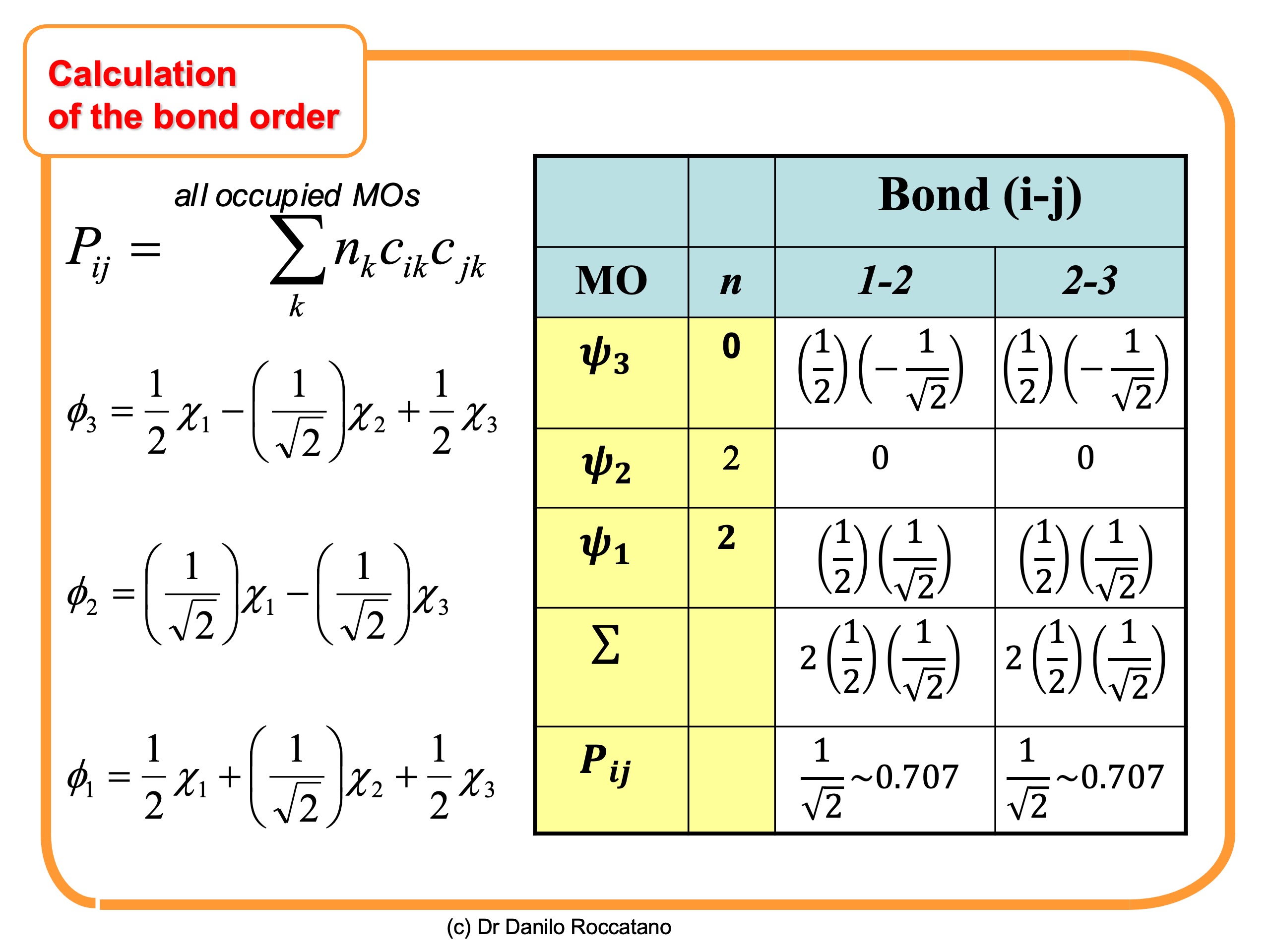
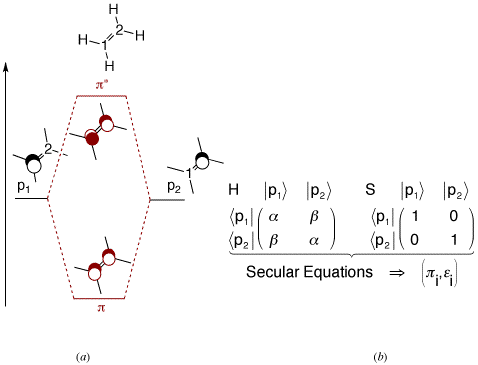
computational chemistry - Tight binding model calculation with Extended Huckel Approximation - Computational Science Stack Exchange