PDF) ESOPEC: Prospective randomized controlled multicenter phase III trial comparing perioperative chemotherapy (FLOT protocol) to neoadjuvant chemoradiation (CROSS protocol) in patients with adenocarcinoma of the esophagus (NCT02509286)
![PDF] Cross-trial prediction of treatment outcome in depression: a machine learning approach. | Semantic Scholar PDF] Cross-trial prediction of treatment outcome in depression: a machine learning approach. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6194c942b91634e8b7c340f158ba759b0789ca7d/5-Figure2-1.png)
PDF] Cross-trial prediction of treatment outcome in depression: a machine learning approach. | Semantic Scholar
![PDF] Accuracy of Detecting Residual Disease After Cross Neoadjuvant Chemoradiotherapy for Esophageal Cancer (preSANO Trial): Rationale and Protocol | Semantic Scholar PDF] Accuracy of Detecting Residual Disease After Cross Neoadjuvant Chemoradiotherapy for Esophageal Cancer (preSANO Trial): Rationale and Protocol | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/630bc6b873bf8304e24a6ac1260e289c3deac5d2/5-Figure1-1.png)
PDF] Accuracy of Detecting Residual Disease After Cross Neoadjuvant Chemoradiotherapy for Esophageal Cancer (preSANO Trial): Rationale and Protocol | Semantic Scholar

Neoadjuvant treatment strategy for locally advanced thoracic esophageal cancer - Mayanagi - 2019 - Annals of Gastroenterological Surgery - Wiley Online Library
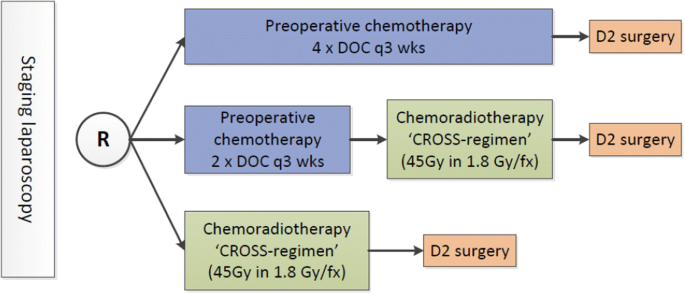
CRITICS-II: a multicentre randomised phase II trial of neo-adjuvant chemotherapy followed by surgery versus neo-adjuvant chemotherapy and subsequent chemoradiotherapy followed by surgery versus neo-adjuvant chemoradiotherapy followed by surgery in ...
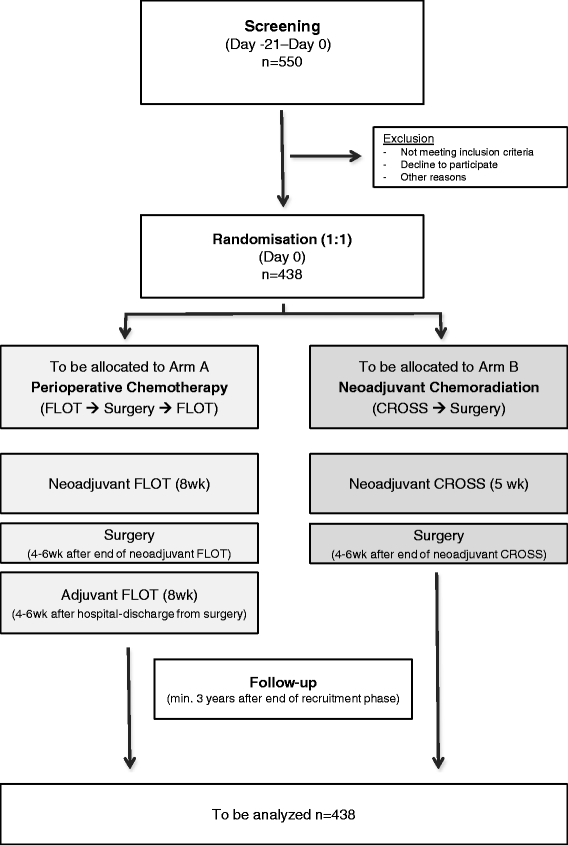
ESOPEC: prospective randomized controlled multicenter phase III trial comparing perioperative chemotherapy (FLOT protocol) to neoadjuvant chemoradiation (CROSS protocol) in patients with adenocarcinoma of the esophagus (NCT02509286) | BMC Cancer | Full ...

Journal of Clinical Oncology on X: "Survival benefit of preoperative chemoradiotherapy for patients with esophageal cancer persists - 10-year outcome of the CROSS trial https://t.co/s02sMVpYhI #esocsm #JCO @ErasmusMC @bmeyck https://t.co/2JwV0iOvoB" / X
Total neoadjuvant therapy in oesophageal and gastro-oesophageal junctional adenocarcinoma | British Journal of Cancer
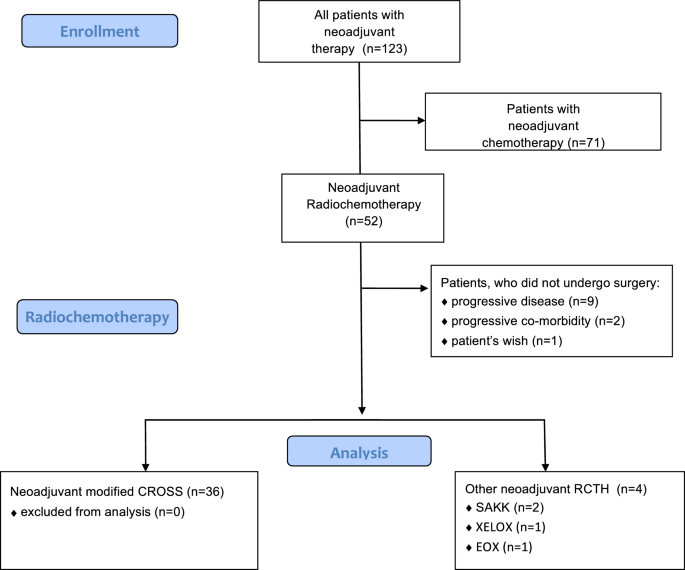
Modification of preoperative radiochemotherapy for esophageal cancer (CROSS protocol) is safe and efficient with no impact on surgical morbidity | Strahlentherapie und Onkologie
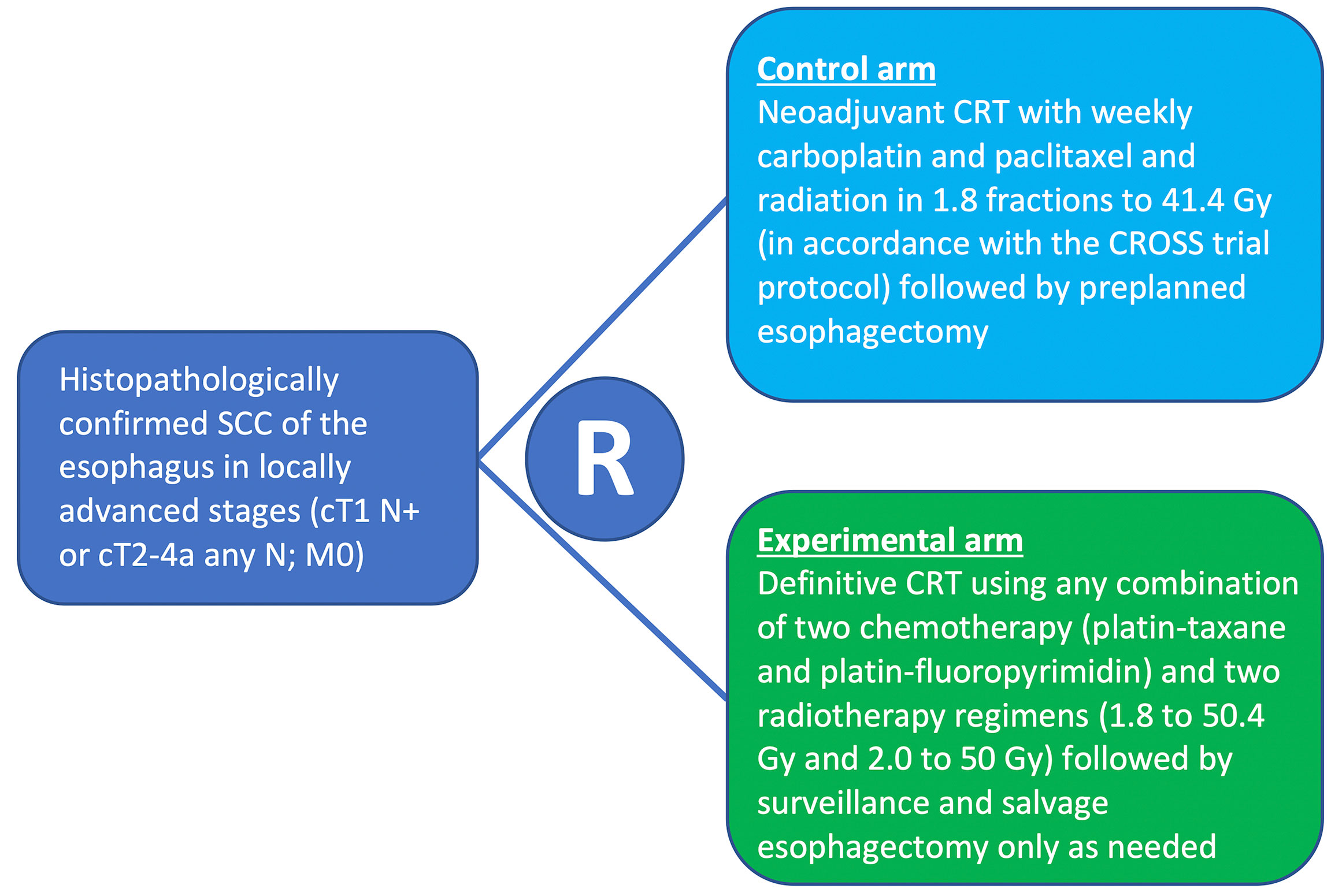
Frontiers | Neoadjuvant Chemoradiotherapy and Surgery for Esophageal Squamous Cell Carcinoma Versus Definitive Chemoradiotherapy With Salvage Surgery as Needed: The Study Protocol for the Randomized Controlled NEEDS Trial

Real-world Scenario: CROSS Regimen as Preoperative Therapy for Oesophageal Squamous Cell Carcinoma | Journal of Gastrointestinal Surgery

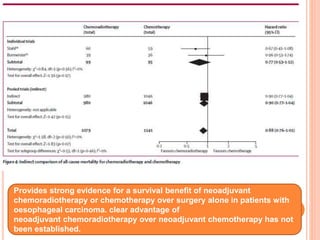
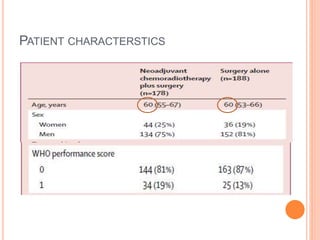
Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): long-term results of a randomised controlled trial - The Lancet Oncology

Bennouna Jaafar on X: "From the CROSS trial to CheckMate 577. Need to have OS data in CM577. longer follow-up. And come back more precisely to CPS-PD-L1 data. https://t.co/aUy1dDuxQ1" / X
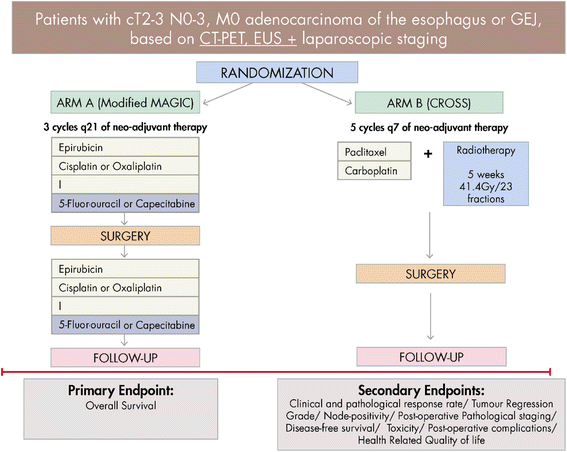
ICORG 10-14: NEOadjuvant trial in Adenocarcinoma of the oEsophagus and oesophagoGastric junction International Study (Neo-AEGIS) | BMC Cancer | Full Text