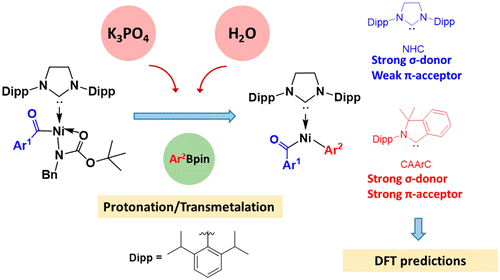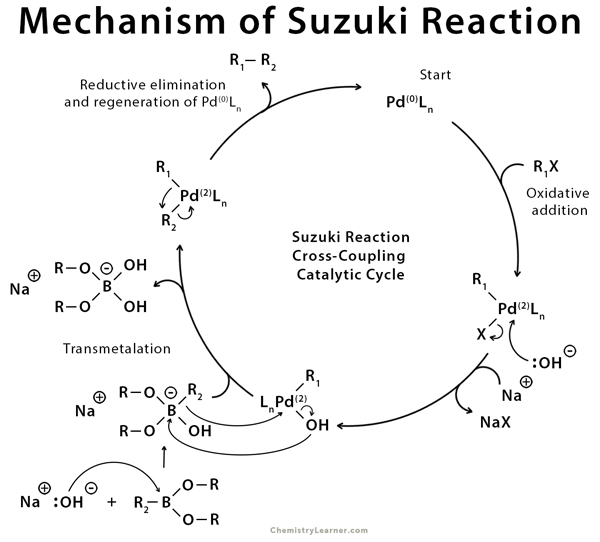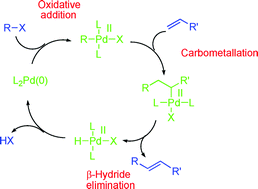
The Heck–Mizoroki cross-coupling reaction: a mechanistic perspective - Organic & Biomolecular Chemistry (RSC Publishing)
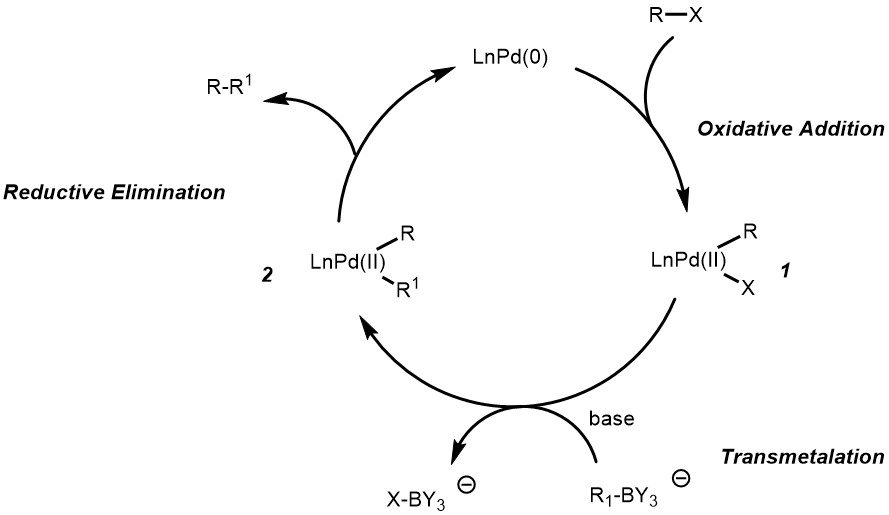
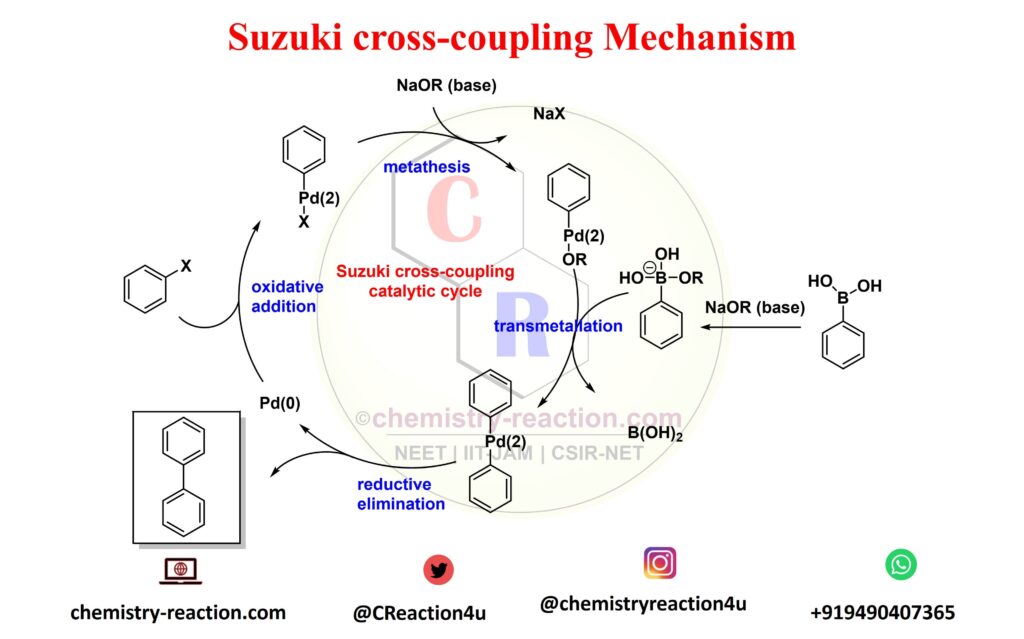
Suzuki–Miyaura cross-couplings for alkyl boron reagent: recent developments—a review | Future Journal of Pharmaceutical Sciences | Full Text
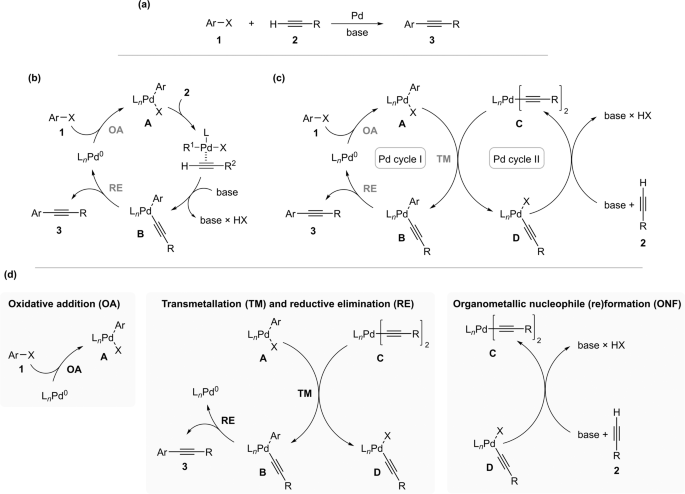
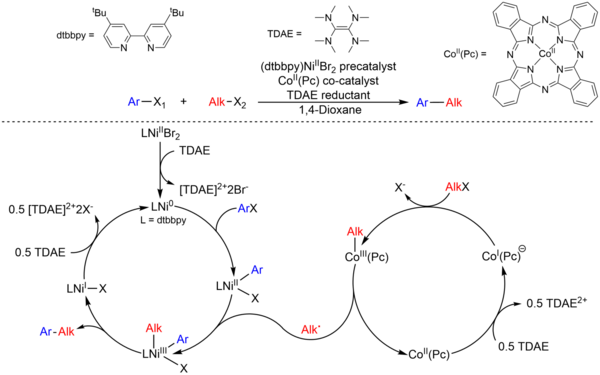
Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps | Communications Chemistry

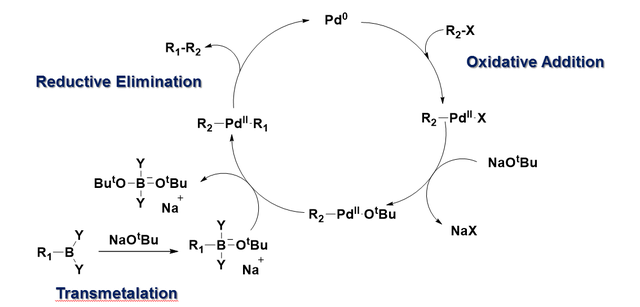
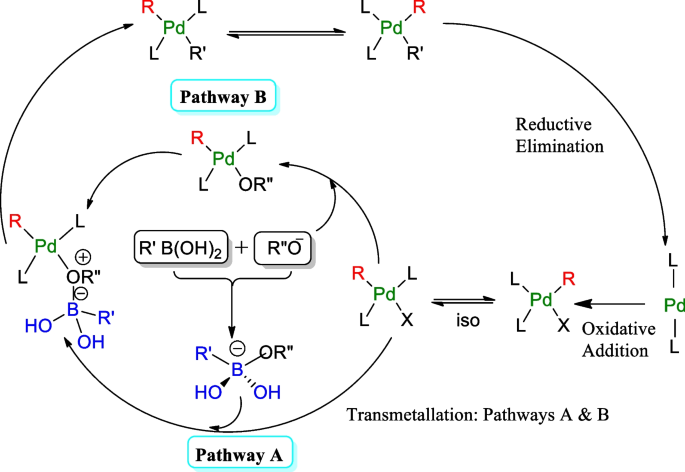
Figure 7 from Transition-metal-catalyzed Suzuki-Miyaura cross-coupling reactions: a remarkable advance from palladium to nickel catalysts. | Semantic Scholar
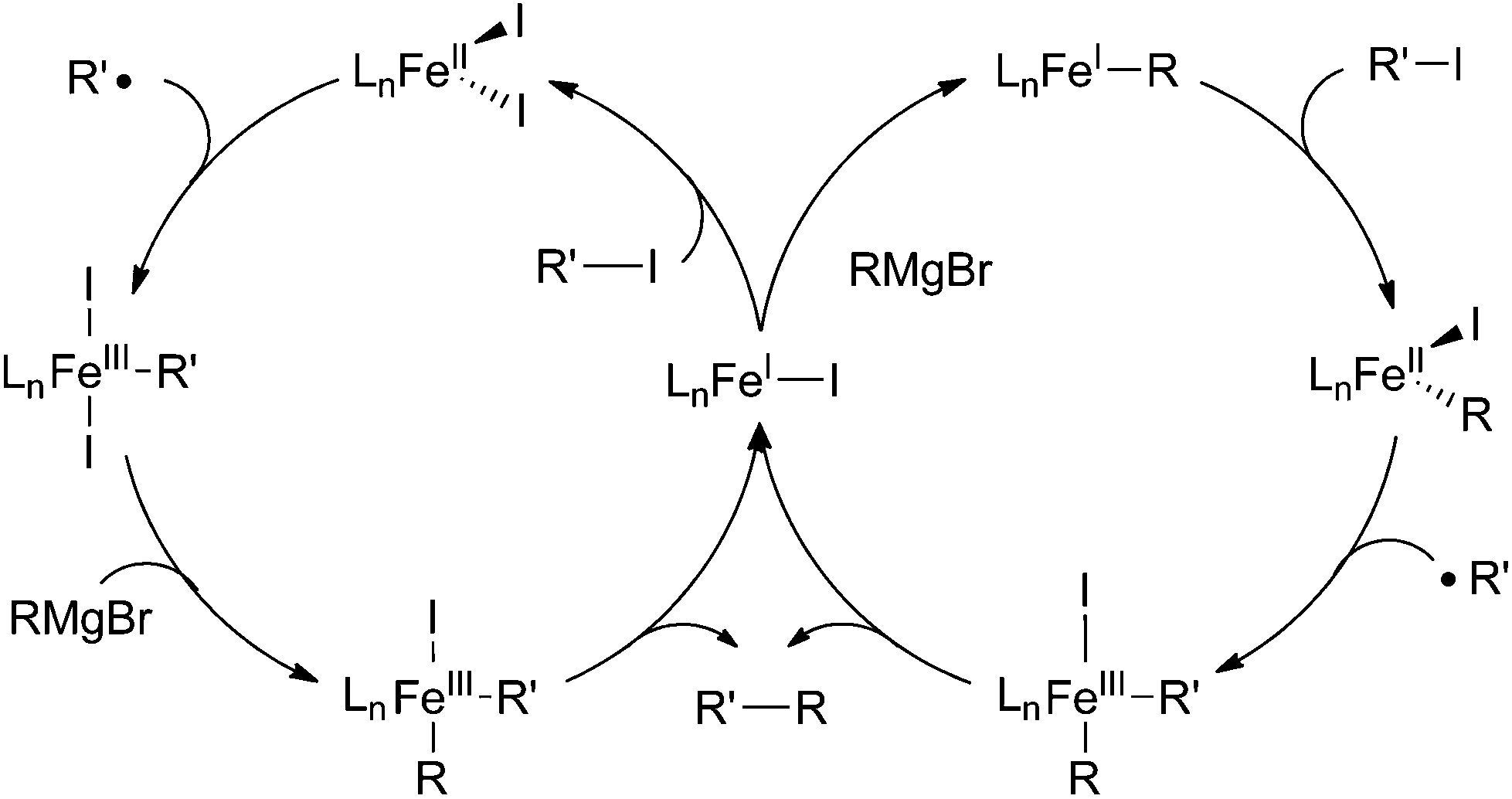
Recent advances in iron-catalysed cross coupling reactions and their mechanistic underpinning - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C5QI00295H
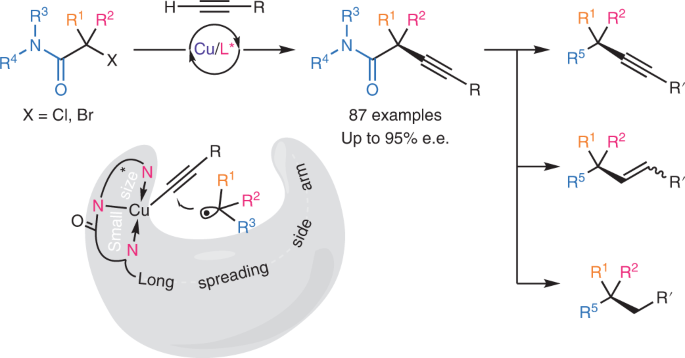
Mechanism-based ligand design for copper-catalysed enantioconvergent C(sp3)–C(sp) cross-coupling of tertiary electrophiles with alkynes | Nature Chemistry
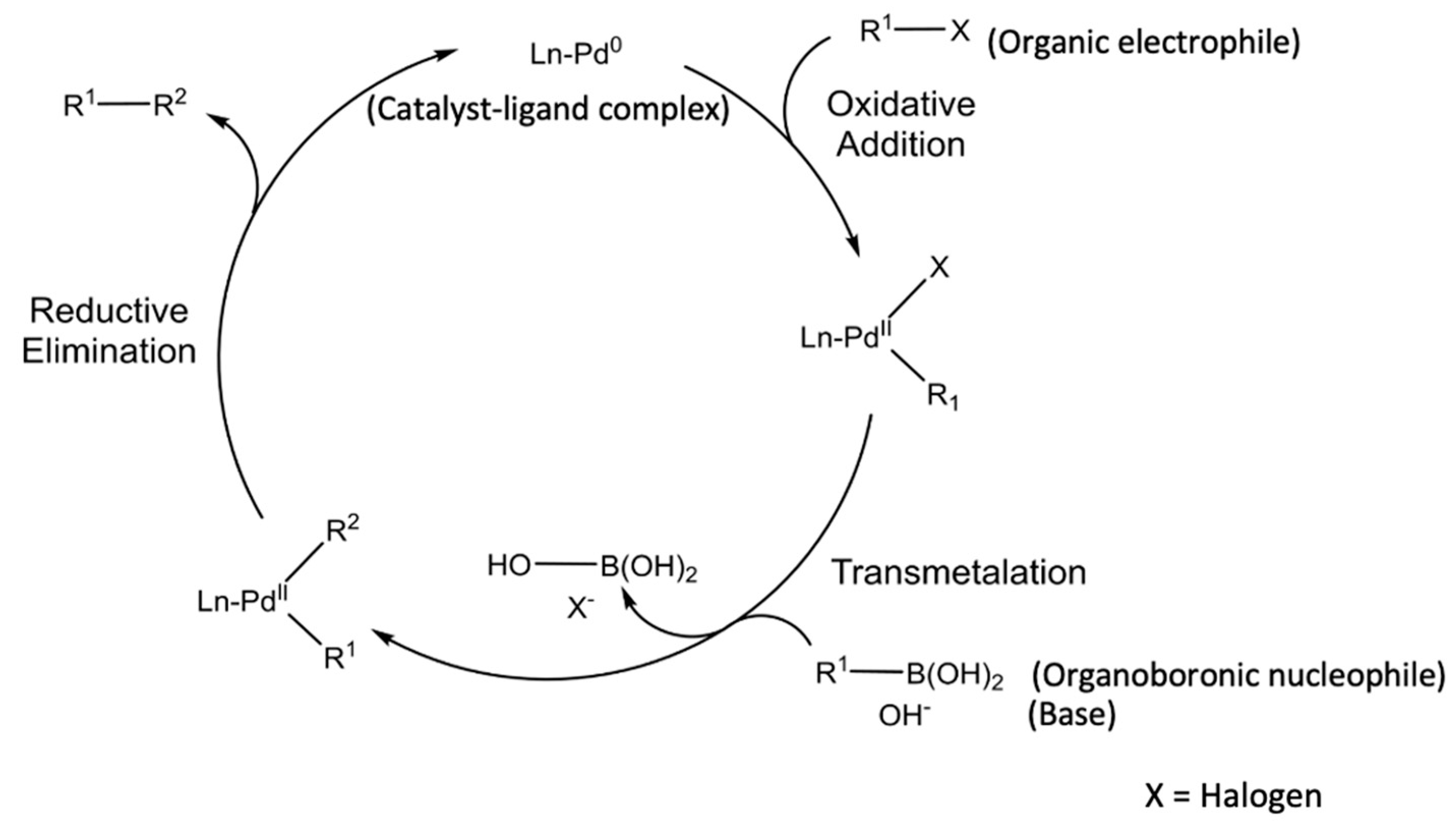



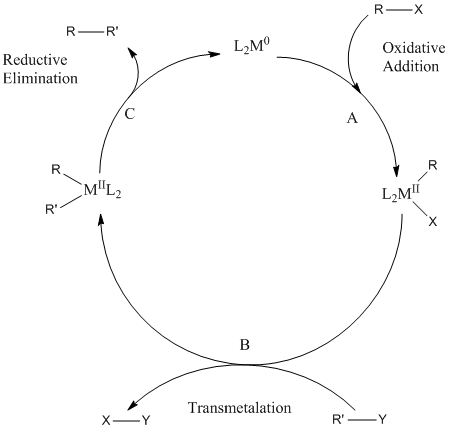
![PDF] Mechanisms of Nickel-Catalyzed Cross-Coupling Reactions | Semantic Scholar PDF] Mechanisms of Nickel-Catalyzed Cross-Coupling Reactions | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3297cc29ee21e706a719bd5da81dca59c2ac6ab2/7-Figure4-1.png)