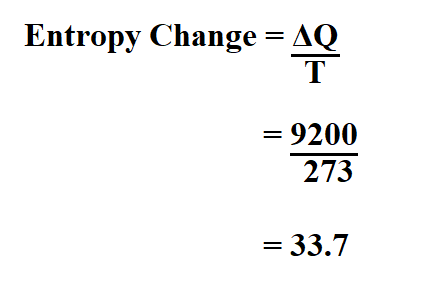calculate the entropy change when 1 mole of an ideal gas is allowed to expand isothermally at 315k and pressure 5atm to 2.5atm - EduRev Class 11 Question
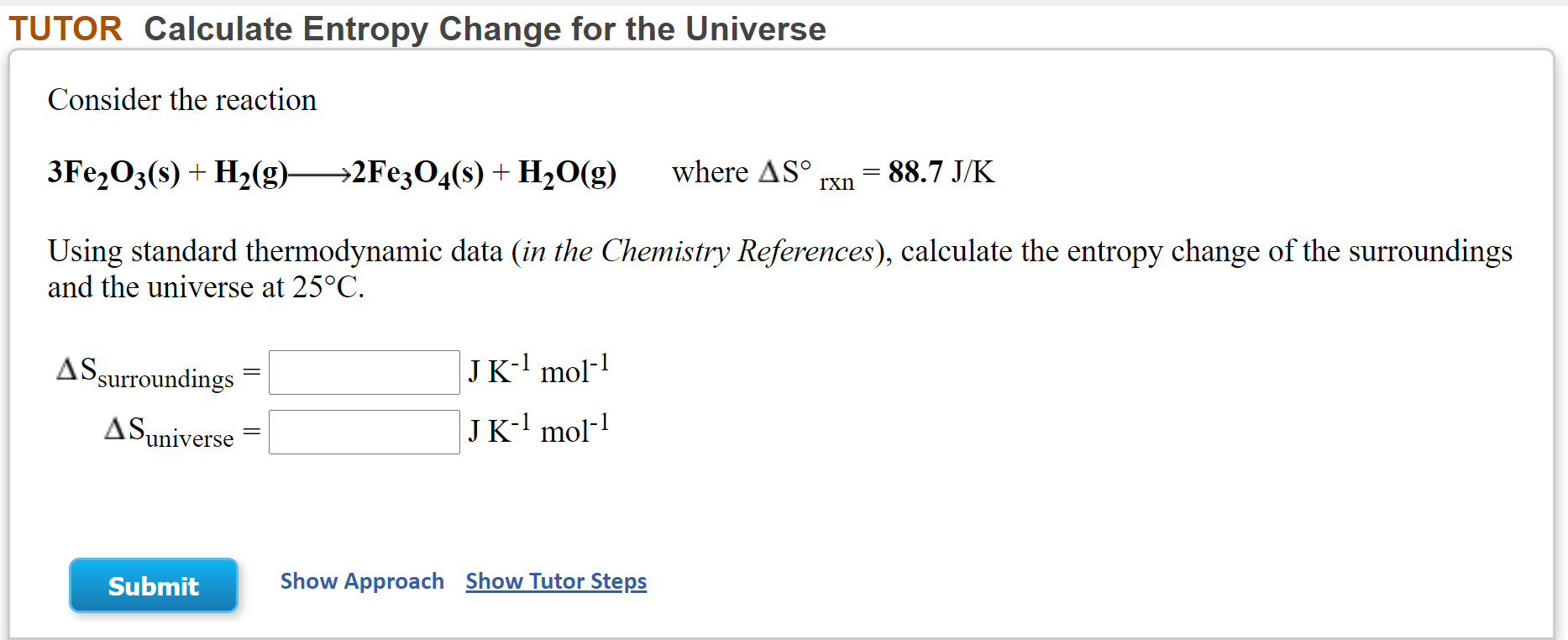
Calculate the entropy change in the system. and in the surroundings and the total entropy change in the universe when during - Sarthaks eConnect | Largest Online Education Community

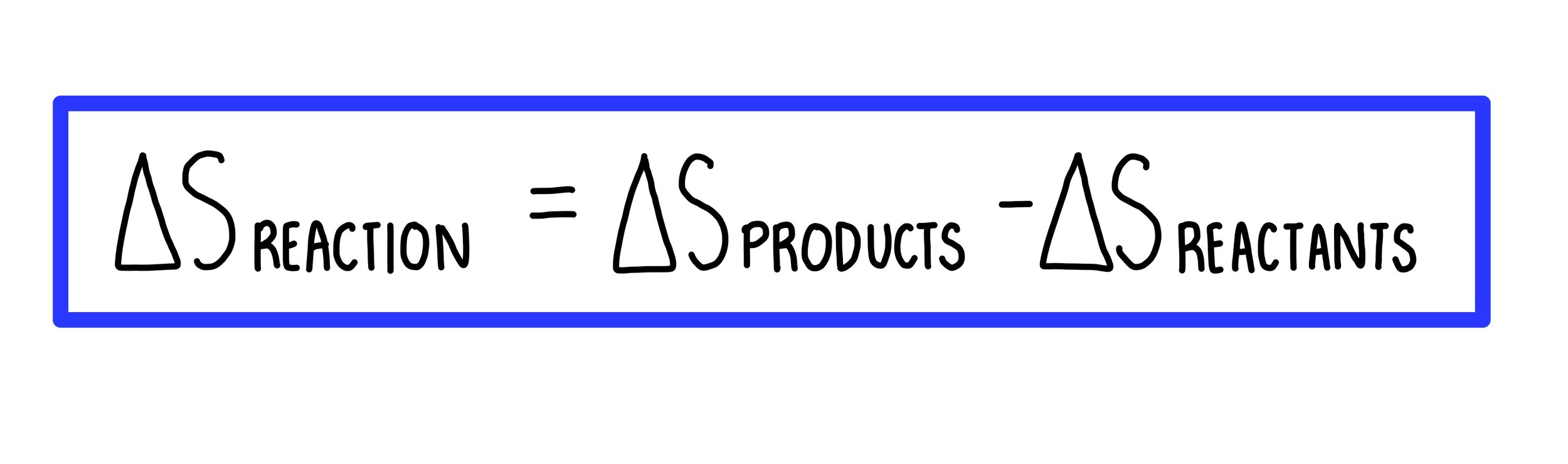
Can you calculate the entropy change when one mole of an ideal gas expands to double its original volume? - Quora
The entropy of vaporization of benzene is 85 JK^ 1mol^ 1. When 117 g benzene vaporizes at its normal boiling point then the entropy change of surrounding is : (1) 85 JK^ 1 (2) 85 × 1.5 JK^ 1 (3 ) 85 × 1.5 JK^ 1 (4) None of these .