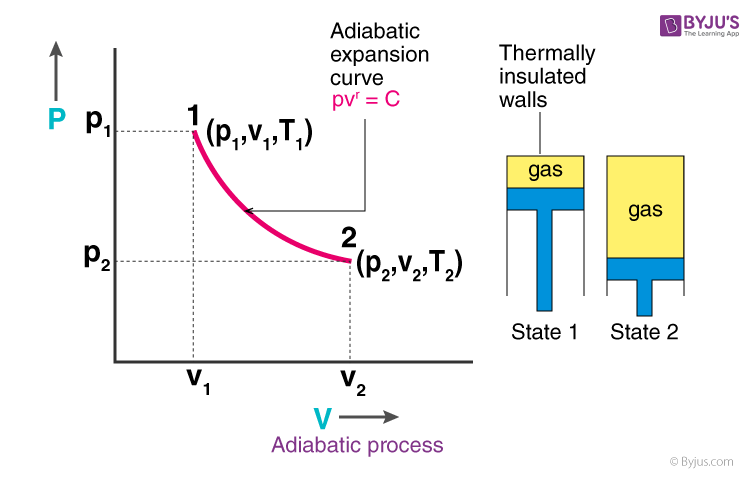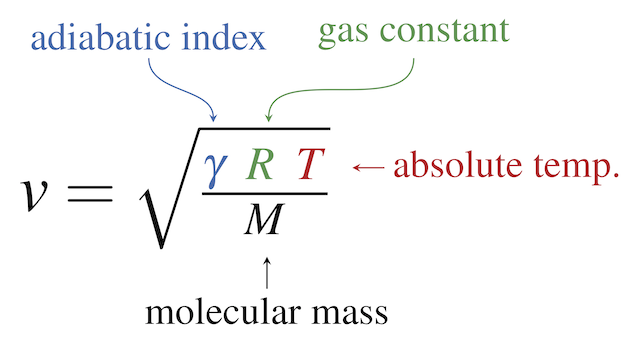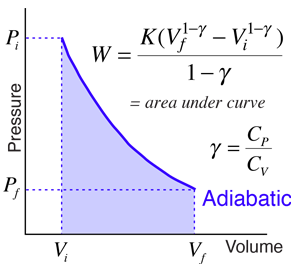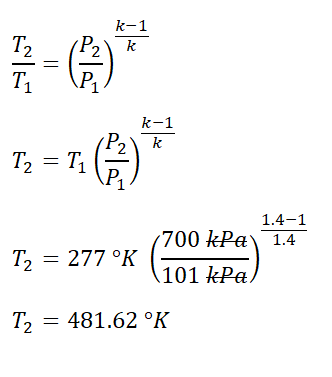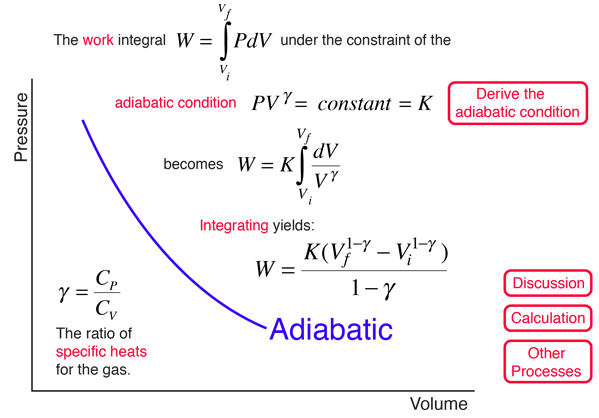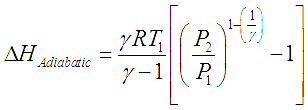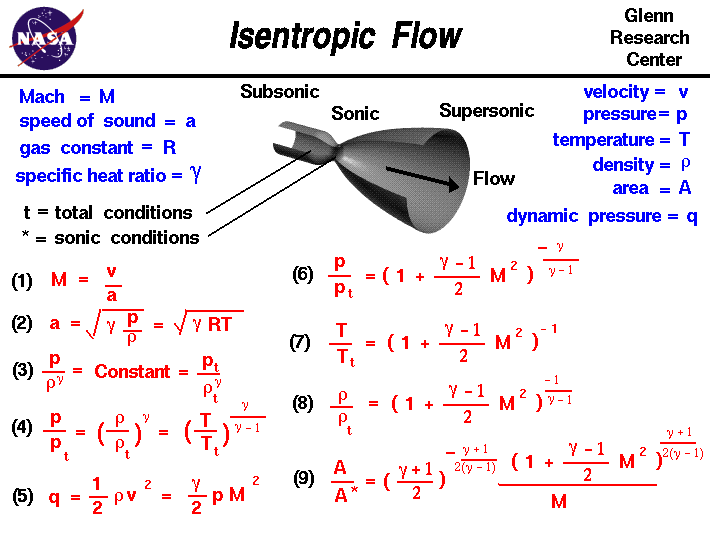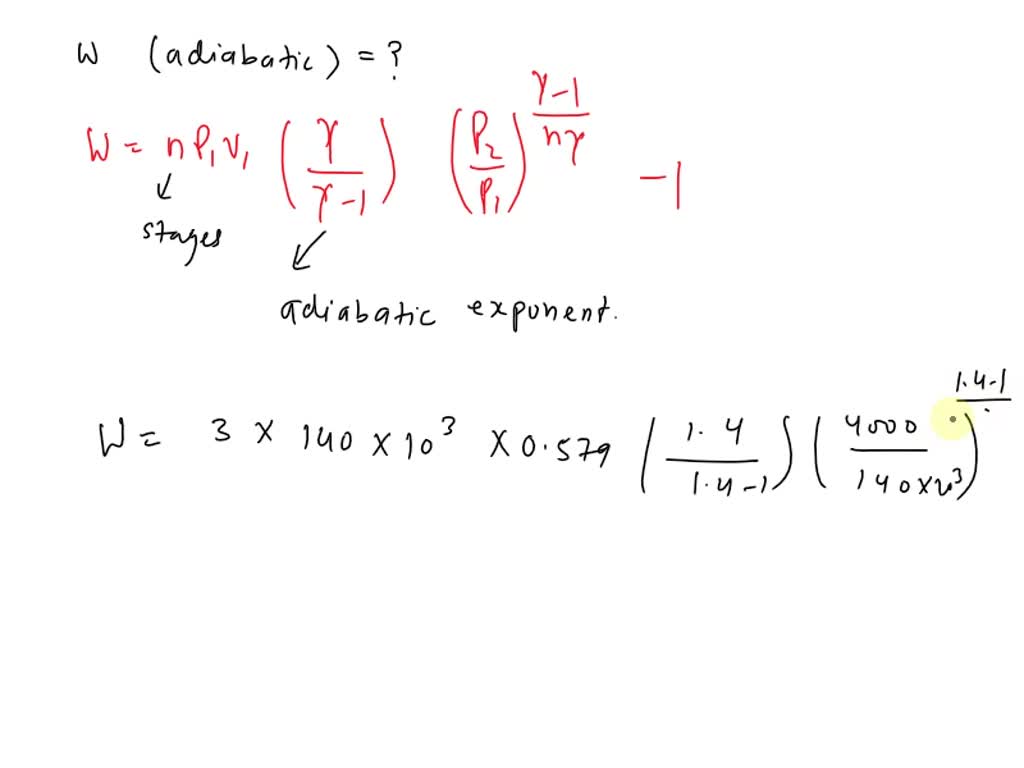
SOLVED: A three-stage compressor is required to compress air from 140 kN/m2 and 283 K to 4000 kN/m2. It may be assumed that the compression is adiabatic and interstage cooling is provided

9. EN S UIVN-0.08 UDMIL. A monoatomic ideal gas undergoing irreversible adiabatic compression from 4L to IL against I bar pressure. Calculate the enthalpy change of gas. [Given : 1 bar-L =
Suppose an irreversible adiabatic compression at constant external pressure (bigger than internal pressure) How can we calculate the change in internal energy of the system? - Quora